The following are the main working groups that, on an informal basis, have been promoted by non-institutional actors who have asked Cittadinanzattiva/ACN to participate:
-
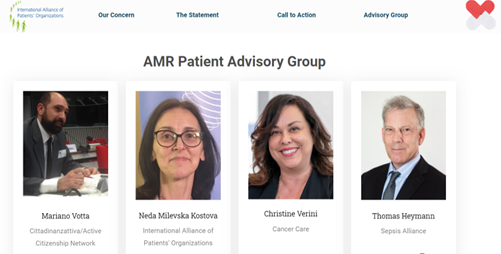
Since 2021: In line with its long-term commitment on Healthcare-Associated Infections (HAIs) and Antimicrobial Resistance (AMR), ACN is part of the “AMR Patient Alliance Advisory Group”, an initiative promoted by the International Alliance of Patients’ Organisations (IAPO), with whom ACN has officially formalized a Memorandum of Understanding.
-
Since 2021: In line with its long-term commitment at the national and European levels on vaccination, part of the “Steering Group on Influenza Vaccination”, a coalition promoted by Vaccines Europe comprised of policymakers, patient group representatives, patient organisations, clinicians, and industry representatives active on influenza vaccination since years with the objective to promote an increased uptake of influenza vaccination across Europe. As part of the group, on the occasion of the EU Flu Day 2021, Active Citizenship contributed to the promotion of a Joint Statement on the importance of healthcare professionals (HCP) vaccination with the aim to highlight the importance of strengthened efforts at the EU level to ensure healthcare professionals across Europe are protected in the very unpredictable influenza season ahead. To read the document, please click here.

-
Since 2021: ACN has been invited by the International Federation on Ageing (IFA), with whom ACN has signed an official Memorandum of Understanding, to join the “Vaccination Advocacy Toolkit Advisory Group Meeting”, an international stakeholder group promoted by IFA on January 2021 to foster discussion and determination on the elements necessary in the Vaccination Advocacy Toolkit (VAT) to influence and stimulate vaccination policies and practice in the most at-risk populations.

-
Since 2020: In line with its long-term commitment at the national (Italian) level on the topic of diabetes and obesity, Cittadinanzattiva is active in the field also at an European level through its EU branch ACN, as testified by its participation in the “Obesity Policy Engagement Network (OPEN)”, promoted by the European Association for the Study of Obesity (EASO), with whom ACN has officially signed a Memorandum of Understanding.

-
Since 2019: In line with its long-term commitment on Healthcare-Associated Infections (HAIs) and Antimicrobial Resistance (AMR), ACN has been invited to join the “Global Anti-Infectives (AI) and Antimicrobial Resistance (AMR) Patient Advocacy Advisory Summit”, promoted by Pfizer, to discuss, together with leaders of other patient advocacy groups coming from all the world, key issues affecting anti-infective therapies and antimicrobial resistance (AMR). To read a brief summary, click here.

-
Since 2019: ACN has been invited to join the “AMR Stakeholder Network”, an initiative promoted by the European Public Health Alliance (EPHA) in 2019, bringing together more than 60 leading organisations and individuals committed to tackling Antimicrobial Resistance (AMR) at national, regional and European level, covering all dimensions of the ‘One Health’ approach. Since 2019, also Member of the “MEP Interest Group to tackle AMR”, launched on 21 November 2019 and supported by the AMR Stakeholder Network.

-
Since 2019: As a follow up of the officially formalized agreement with the Federation of European Academies of Medicine (FEAM), ACN has joined the FEAM European Biomedical Policy Forum, a platform for discussion on key policy issues for the biomedical community. It aims to bring together representatives from academia, research charities, industry, European and national trade associations and professional bodies, regulators, public health bodies, and patient and consumers groups.

-
Since 2019: Part of the EU Health Coalition, a multi-stakeholder initiative promoted by European Federation of Pharmaceutical Industries and Associations (EFPIA) and looking at mapping the future of healthcare in Europe in order to make the most of the innovation at our fingertips, at the role of the European Union in addressing the challenges we face and critically at how can different sectors converge to deliver the best outcomes for patients in Europe. To read the statement done by the ACN Director, click here.
-
Since 2018: Active Citizenship Network is Knowledge Partner of “All Policies for a Healthy Europe”, an intersectoral initiative promoted by companies, that brings together a diverse group of NGOs, think-tanks, associations, companies, and individuals. The mission is to put citizens’ health & well-being at the heart of European policymaking. To know more, click here.
-
Since 2014: Cittadinanzattiva, through its EU branch Active Citizenship Network, is part of “Fight the Fakes” (FTF), a multi-stakeholder non-profit association that aims to raise awareness and influence change about the proliferation of substandard and falsified medicines. For more info, click here and here.


-
Since 2014: Member of the EuropaBio Patients-BioIndustry Exchange Forum (EuropaBio Patient BioForum), a platform for the exchange of views and expertise between patient organisations. Through it, the input and views from patient organisations on scientific, regulatory and policy issues are discussed and taken into account to better support our contributions to the healthcare policy debate in the EU.




